Our Thinking
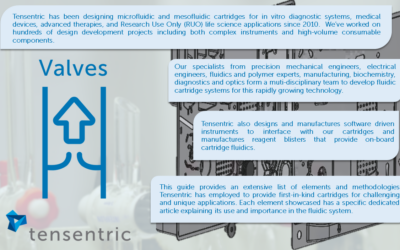
Valves- A Guide to Meso and Microfluidics
Valves Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for diagnosing diseases,...
Containment of On-Board Reagents- A Guide to Meso and Microfluidics
Containment of On-Board Reagents Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for...
Vents- A Guide to Meso and Microfluidics
Vents Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for diagnosing diseases,...
Pumps- A Guide to Meso and Microfluidics
Pumps Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for diagnosing diseases,...
Materials- A Guide to Meso and Microfluidics
Materials Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for diagnosing diseases,...
Manufacturing- A Guide to Meso and Microfluidics
Manufacturing Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for diagnosing diseases,...
Filters- A Guide to Meso and Microfluidics
Filters Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for diagnosing diseases,...
Mixing- A Guide to Meso and Microfluidics
Mixing Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for diagnosing diseases,...
Sensing Capabilities- A Guide to Meso and Microfluidics
Sensing Capabilities Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for diagnosing...
Electrodes and other Elements- A Guide to Meso and Microfluidics
Electrodes and other Elements Tensentric instruments provide efficient and hands-off analysis of lab samples, replacing complex, time-consuming and potentially error-prone steps crucial for...
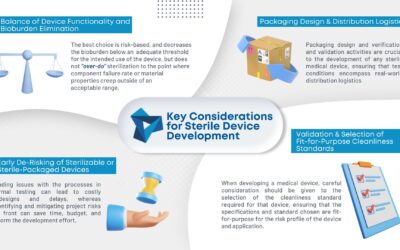
KEY CONSIDERATIONS FOR STERILE DEVICE DEVELOPMENT
As medical device consumables become more complex, so does manufacturing them. Sterilization is becoming an increasing concern in the industry. To ensure the proper sterilization is in place, there...
WHAT IS HUMAN FACTORS ENGINEERING AND WHY IS IT IMPORTANT?
Medical devices and IVD systems are developed with the purpose of assisting health care providers diagnose and treat patients to overcome sickness or disease to, in the end, improve their quality of...
BEYOND USABILITY: DESIGNING CLIA WAIVED DEVICES
Are you ready for the Clinical Laboratory Improvement Amendments waiver? With the large market for laboratory devices growing fast, the benefits of designing for the waiver are even more important...
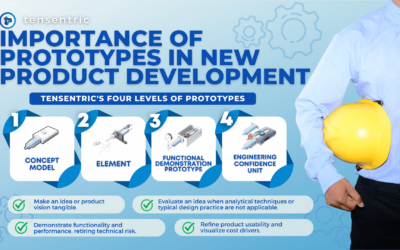
Importance of Prototypes in New Product Development
In NPD, a prototype is a representation of a product idea intended to learn something about it.
Microsoft Blazor: Finally, a Cross-Platform GUI Solution for Medical Devices
Every product development effort containing embedded software must answer the question early on “Which operating system should the device run?”
IVD System Development: Don’t Forget the Assay
Iterating product design while keeping biochemical system interfaces at the forefront de-risks key technical challenges earlier and accelerates device development.
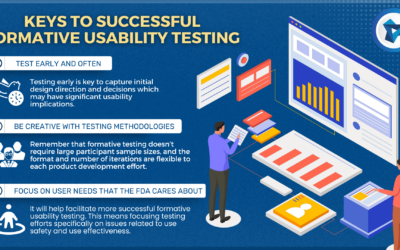
Formative Usability Testing in Medical Device Development
Imagine a medical device development team has done preliminary analyses on who will be using the device.
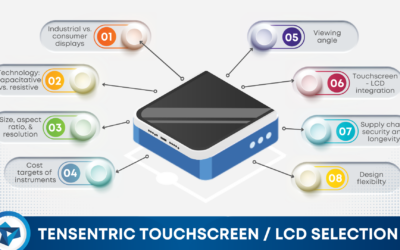
Choosing Touchscreen LCDs for your Medical Device Design
There are many features that drive LCD and touchscreen selection for a device in any industry, but these decisions become more restricted in the medical device space.
In-House Rapid Prototype Fabrication Capabilities
Tensentric offers in-house machining, 3D printing, and other prototyping options to accelerate the design and development cycle with rapid iteration.
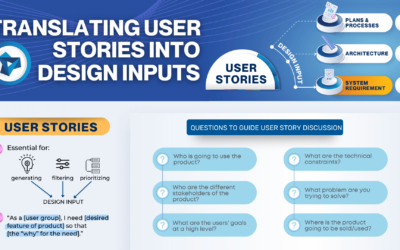
Translating User Stories into Design Inputs
The process of defining user stories, asking questions about the intended user group(s), and identifying the user’s needs helps define the “why” behind each design input, resulting in a minimum viable product.
Designing Medical Devices with Cellular IoT
Connecting medical devices to the cloud provides tremendous advantages, and using Cellular IoT technology may be the only way to make the connection.
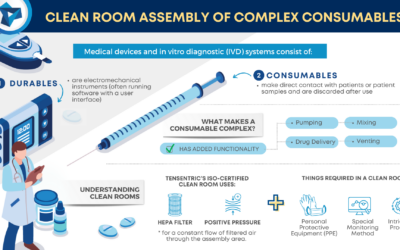
Clean Room Assembly of Complex Consumables
For complex consumables requiring assembly in a certified clean room, Tensentric’s experienced manufacturing team and FDA registered, ISO 13485 facility is here to help bring your product to market.
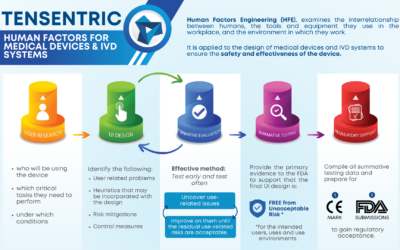
Human Factors for Medical Devices & IVD Systems
A thorough and robust Human Factors process is essential and required for medical device and in vitro diagnostic system design.
The Art of Usability Test Moderation
With some hands-on practice, tips and guidance, you can master this art and science of moderation and collect high-quality data.
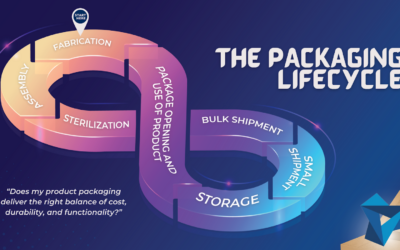
Medical Packaging Design: Thinking Outside the Box
When designing a new medical, diagnostic, or life science device, the safe and effective delivery of product is just as important as its safe and effective use. Often there are several levels of...
Battery Selection Analysis for Medical Device Design
When designing of a battery-operated medical device, selecting the battery cannot be postponed.
Designing Connected Devices with Bluetooth Low Energy
When developing wireless communication solutions, three things matter – security, performance, and cost of implementation.
Low-Cost RFID Reader Design for Embedded Medical Devices
For many high volume medical devices, a pre-certified RFID module can be cost-prohibitive.
Plastic Part Tooling Strategies for Medical Device Development
Choosing a fabrication method for medical products with plastic parts carry added complexity due to the materials, precision needed, and regulatory environment.
Securing Firmware in Embedded Devices
Implementing Secure Boot and Secure Firmware Updates in embedded devices ensures trusted code execution.
Design for EMC Test: An Interdisciplinary Approach
Designing a medical device to meet the IEC 60601-1-2 4th edition Electromagnetic Compatibility (EMC) standards is notoriously difficult. Making matters worse, the details of what goes on during EMC...
Wireless Power: How Small Can We Go?
Wireless power is ideal for charging sealed, battery-operated, handheld medical devices, and the technology can be reduced in size to in the smallest of devices.