Designing a Robust Security Solution
Cybersecurity implementation and support in the total product lifecycle
A strong cybersecurity framework is created starting at Phase Zero. New product design benefits the most by dedicating effort in Phase Zero in reviewing and refining security needs.
In Phase Zero, we explore the right tools and technologies to secure your system, ensuring we meet the needs of both patients and business. Early vulnerability analysis leads to a focused threat model, which guides the refinement of the security framework and defines actionable requirements.
Our approach in integrating security early, and throughout the standard software design controls, ensures that Tensentric is designing a panoramic security foundation for our clients.

While creating a security framework from Phase Zero is ideal, many devices reach the market without that foundation. Whether your product was cleared years ago, or you are transitioning design partners, existing designs can gain significant value from Tensentric’s approach to cybersecurity.
We apply the same disciplined process used with new product development: review the security needs of the business and user, explore modern security concepts to meet those needs, perform threat modeling, and create security requirements.
By methodically assessing existing code, we can implement a tailored security framework for your product. This ensures that legacy and existing products gain the same level of rigor, resilience, and regulatory confidence as devices that are built securely from Phase Zero.
Preparing Your Product for Regulated Markets
Safety and security for clinicians, caregivers and patients
We not only design and create these systems, but we also make sure they align with the FDA and other regulatory bodies’ rigorous standards. It’s all about making sure everything is safe, effective, and secure for providers and patients.
We work with our clients to develop a custom design and development plan to pragmatically meet regulatory constraints, from lab devices to cell therapy bioprocessing systems to diagnostic systems and medical devices, in our fully compliant ISO-13485 quality management system.

WORK
WITH US
Interested in how we can help you?
Let’s chat.
Our Thinking
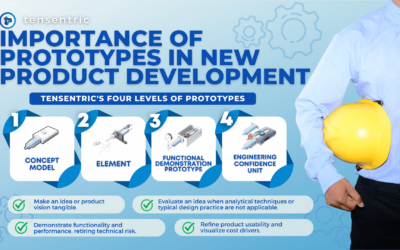
Importance of Prototypes in New Product Development
In NPD, a prototype is a representation of a product idea intended to learn something about it.
Microsoft Blazor: Finally, a Cross-Platform GUI Solution for Medical Devices
Every product development effort containing embedded software must answer the question early on “Which operating system should the device run?”
In-House Rapid Prototype Fabrication Capabilities
Tensentric offers in-house machining, 3D printing, and other prototyping options to accelerate the design and development cycle with rapid iteration.
Team
Our Team Offerings
Human Factors
Concept Ideation & Industrial Design
Mechanical & Electrical Engineering Design
Software & Firmware Development
Design for Reliability & Manufacturability

Assembly and Text Fixture Design & Validation

NPI & Volume Manufacturing
Our Clients
We serve a broad spectrum of clients from early-stage start-ups through large and well-established medical and life sciences OEMs.