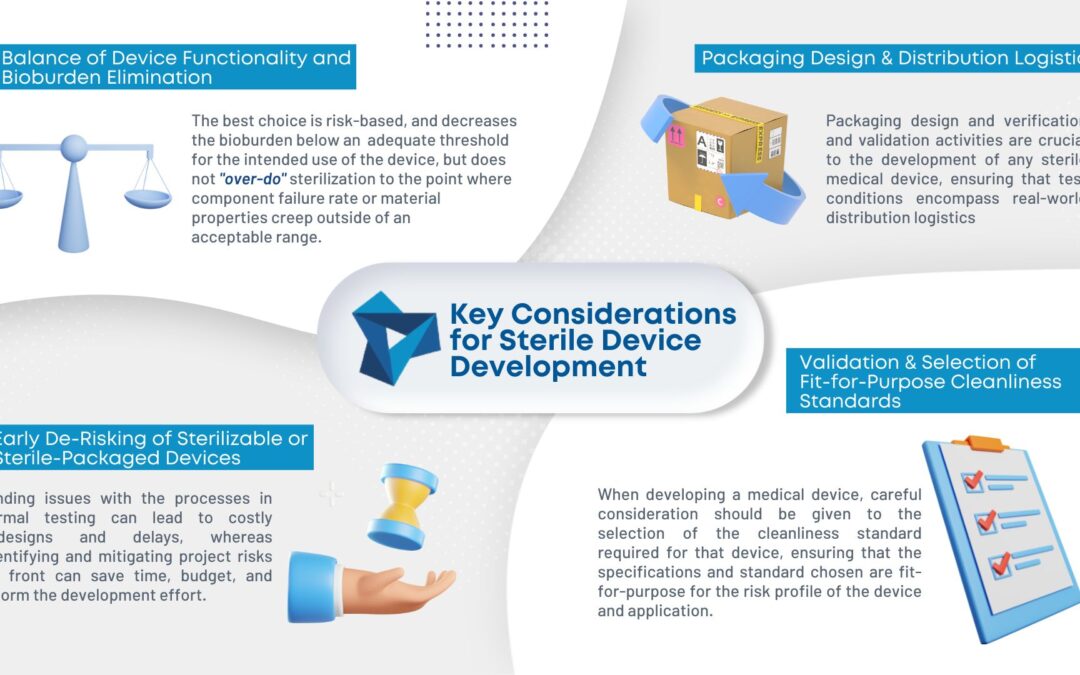
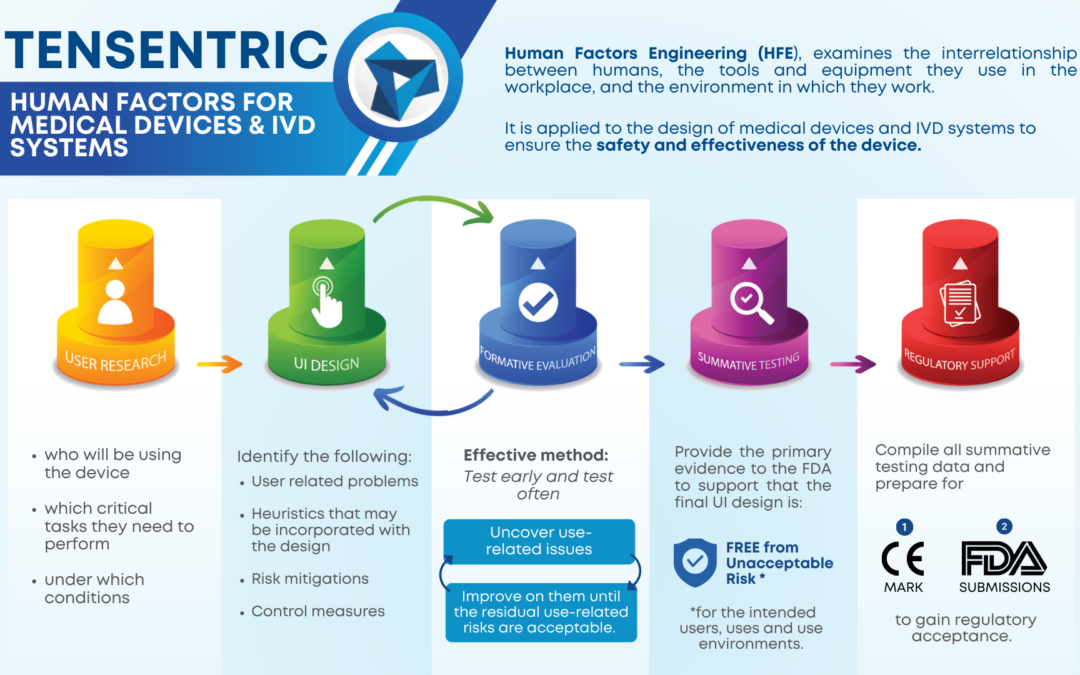
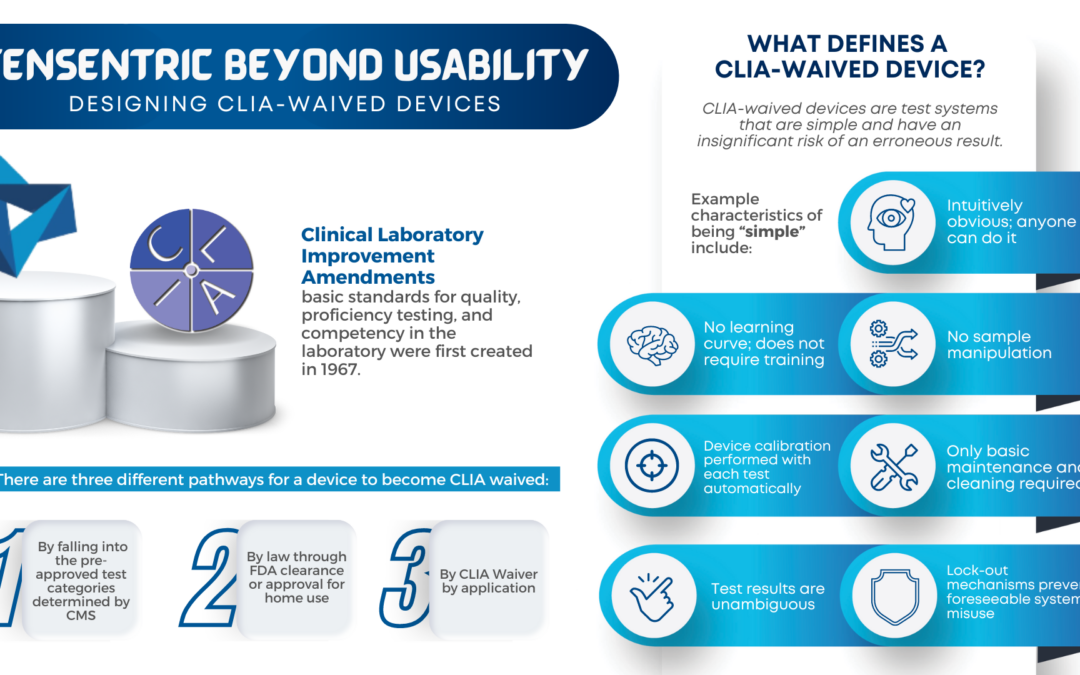
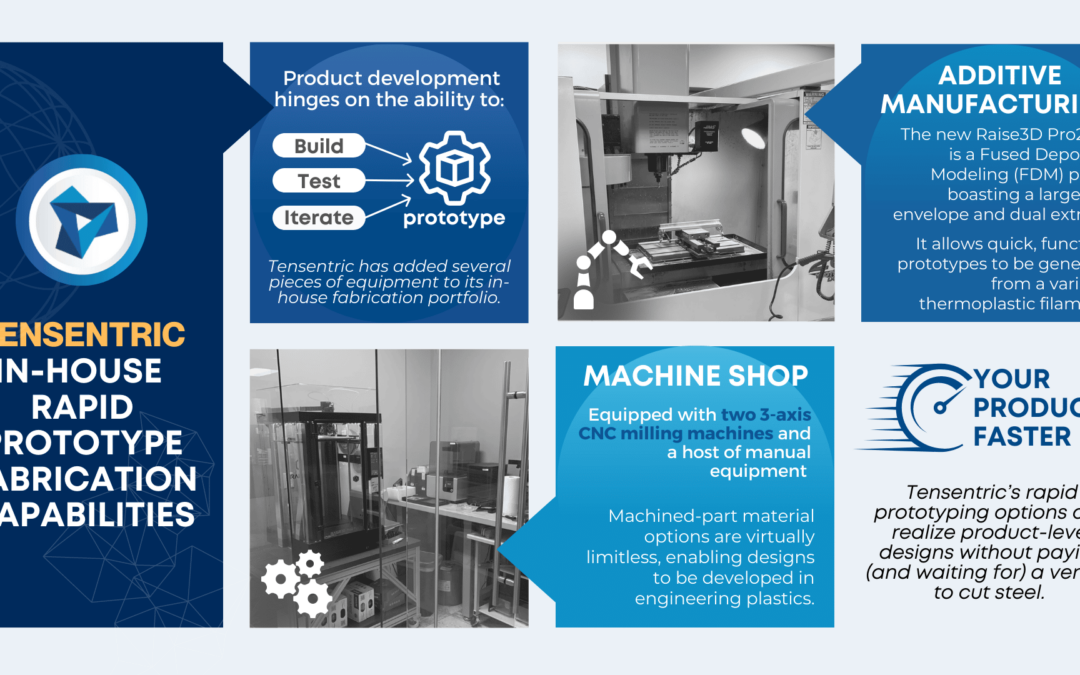
Wireless power has become commonplace for smartphone charging, but did you know that the technology can be reduced in size to fit in all but the smallest handheld medical devices?
Wireless power is ideal for charging sealed, battery-operated,
handheld medical devices
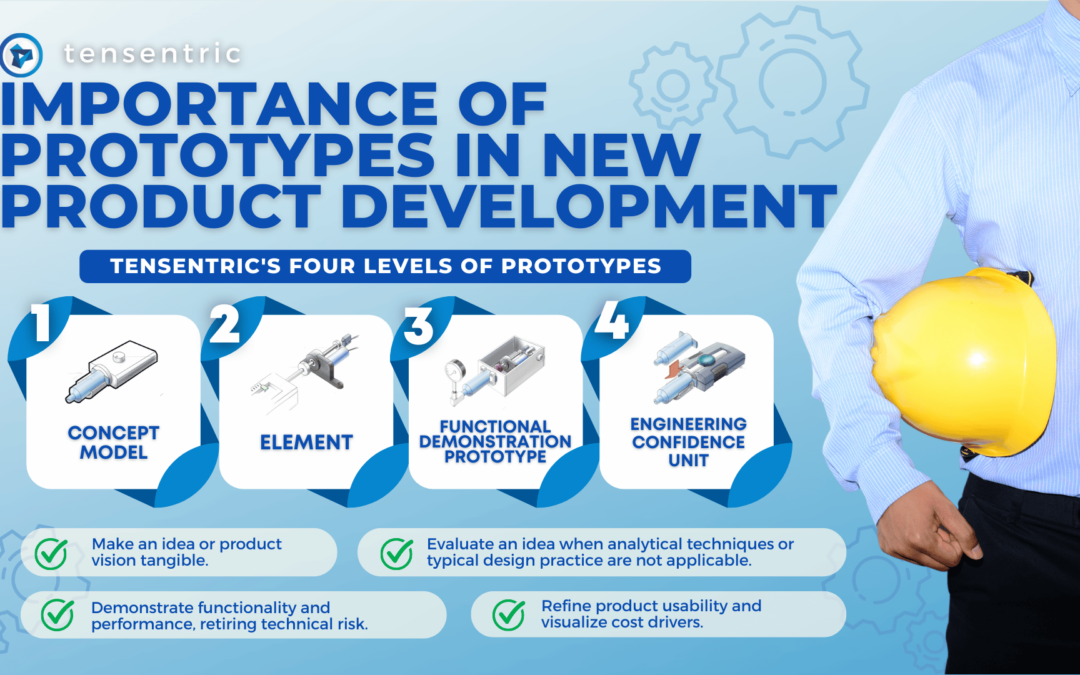
WIRELESS POWER TECHNOLOGY
Wireless power technology uses matched coils to power electronics from a distance without having to plug in a connector. It is ideally suited for charging battery-operated, handheld, medical devices which are sealed to withstand washing and immersion and for which exterior openings need to be avoided to prevent sites that could promote contamination and infection.
There are two wireless charging standards. The Qi (pronounced “chee”) wireless charging standard uses inductive coupling between small distances of around 5mm. The AirFuel Resonant wireless charging standard implements loosely coupled magnetic resonance. After smartphones adopted the Qi wireless charging standard in 2017, Qi became the dominant standard.
WIRELESS POWER DESIGN
A typical wireless power system has 1) a receiver (Rx) in the handheld device that provides energy to charge the battery and 2) a transmitter (Tx) located in a fixed charging base that is connected to a wall power supply. Transmitter power is converted to an AC waveform and then magnetically coupled from the transmitter coil to the receiver coil.
The latest Qi wireless power receiver chips are smaller than 4mm x 4 mm. The receiver chip together with the associated discrete components fit in a 11mm x 11mm area. More challenging is shrinking the receiver and transmitter coils used for inductive coupling.
For a handheld device application, a Qi-compliant receiver and transmitter can be modified for a low-power wireless system by carefully selecting the coil sizes and external component values for the small form-factor design and by modifying the firmware of the wireless power transmitter and receiver chips. Coils can be reduced in size to a point but must still be large enough to transfer power and communicate with the transmitter.
A wireless charging solution company will have several stable, known design recipes for different coil sizes. Recently Tensentric partnered with STMicroelectronics on a very small custom wireless power design for a handheld medical device which used a 10.3mm diameter receiver coil in the handheld device and a 15mm diameter transmitter coil in the charging base.
The best inductive coupling (coupling factor k) occurs when the coils are aligned in the x-y plane, close together (z distance), and matched in diameter. Because the 10.3mm receiver coil was very small, even a slight misalignment between the Rx and Tx coils resulted in a significant reduction in the coupling factor, poor energy-transfer efficiency and sporadic aborted Qi-protocol power transfers. To ensure good energy transfer efficiency and reliable operation, the mechanical design of the charging base had to incorporate a physical means of aiding proper placement of the handheld device on the charging base.
SUMMARY
- Wireless power is ideally suited for charging sealed, battery-operated medical devices.
- The technology can be reduced in size to fit in all but the smallest handheld devices.
- A custom wireless power design requires working closely with a wireless charging solution company to optimize and tune the coil sizes, discrete component values and the firmware of the transmitter and receiver chips.
- Using small receiver coils makes the wireless power design much more sensitive to slight misalignments between the receiver and transmitter coils.
- Incorporating a physical means of coil alignment into the mechanical design of the charging base and the receiver is needed to ensure good energy transfer efficiency and reliable operation with small form-factor designs.